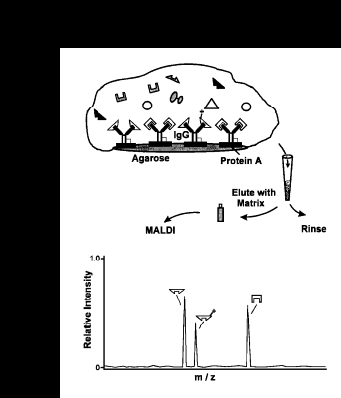
We have developed a new, general method of immunoassay utilizing matrix-assisted laser desorption- ionization time-of-flight mass spectrometry coupled to microscale immunoaffinity capture of target antigens. Mass spectrometric detection of antigens is specific, as antigen signals are observed at characteristic m/z values, permitting their unambiguous determination. This high mass specificity allows the determination of multiple mass resolved antigens in a single assay. Analyte quantitation is possible by using chemically modified variants of the antigen which, along with the antigen, are captured by the single antibody. With proper calibration, the relative signal intensities of the two signals can be used to determine antigen concentration. The general scheme of MSIA is shown below:

Antigens, and chemically modified variants (for quantitative internal references), are captured from solution by use of microliter volumes of MSIA reagent. Following proper incubation, the MSIA reagent-antigen complex is washed repetitively (as shown while retained in a filter pipet tip), and the antigen eluted directly onto a mass spectrometer probe tip with the addition of an aliquot of matrix solution. MALDI time-of-flight mass spectrometry follows, with the antigens (and variants), detected at unique m/z values and quantified with use of the relative ion signals of the antigen and modified variants.
MSIA has been applied to myotoxin a from the venom of the prarie rattlesnake Crotalus viridis viridis . The following figure shows the results of MSIA for mytoxin a present in human whole blood.
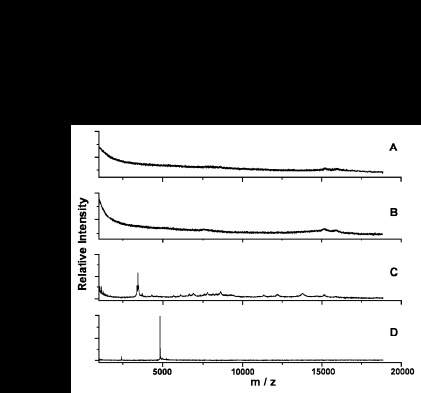
The mass spectrum of human whole blood (A) shows only signals due to hemoglobin. When doped with C.V.viridis venom, the mass spectrum of the blood (B) does not differ significantly from (A). The MSIA spectrum of whole blood is without added venom shown in (C). When MSIA is performed on the doped whole blood, a strong signal for the myotoxin a is seen in (D).
The quantitative response of the MSIA process is demonstrated in the figure below.
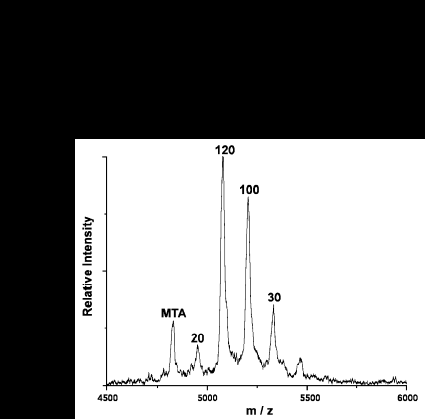
Myotoxin a is modified by successive iodination, resulting in variants each differing in mass by one iodine. Each variant is doped into the analytical sample (which contains the unmodified myotoxin) at a known concentration; in this case at 20, 30, 100, and 120 nM. MSIA is then performed on the sample, and the result is the above spectrum. The intensity of the myotoxin peak falls somewhere between the intensity of the 20 nM and the 30 nM peak, indicating a myotoxin concentration of ~25 nM.
Details of the MSIA procedure can be found in the following publication:
![[*]](images/sphere02.gif) R. W. Nelson, J. R. Krone, A. L. Bieber, P. Williams, Anal. Chem. 67, 1153 (1995)
R. W. Nelson, J. R. Krone, A. L. Bieber, P. Williams, Anal. Chem. 67, 1153 (1995)
![[ <- ]](images/docsleft.gif) Back to Lab Page
Back to Lab Page