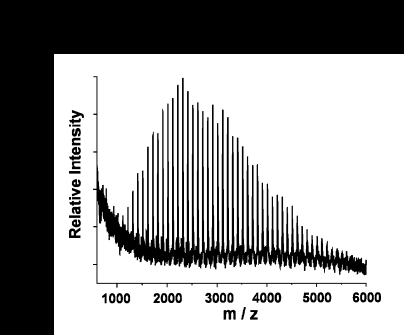
Synthetic (and natural) polymers are another class of compounds that may be analyzed with MALDI-TOF mass spectrometry. Traditionally, molecular weight distributions of polymers have been determined using gel-permeation chromatrography (GPC) or light scattering techniques. With the advances made in the use of matrix materials, time-of-flight mass spectra of polymers have become useful in the determination of polymer distributions. The figure below shows the matrix-assisted time-of-flight mass spectrum of the polymer poly(methylmethacrylate) (PMMA).

Most synthetic polymers ionize via alkali metal ion attachment in the gas phase. The intense peaks in the PMMA spectrum are the result of sodium cationization. This property must be taken into account when choosing solution and matrix conditions in order to successfully obtain mass spectra of polymers.
The matrix pH is another important consideration in MALDI. The effects of changing the pH of the matrix (2,5-dihydroxy benzoic acid, DHB)are shown in the series of spectra below. The matrix is titrated with a solution of potassium hydroxide and aliquots of the resulting titration are taken at the given pH values. The analyte sample contains a mixture of PMMA and the protein alpha-cobratoxin from the venom of the cobra N. n. siamensis.
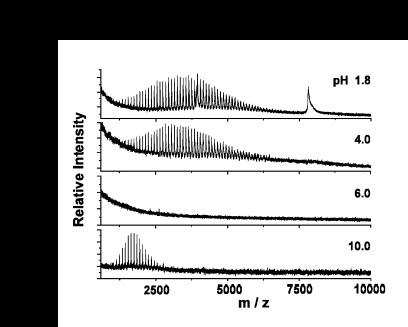
The matrix functions best at its intrinsic pH, as shown in the top spectrum. Intense signals are observed for both the protein and polymer. As the pH is increased, its ability to ionize the protein is lost. In the region of the titration where the pH is changing most rapidly, no signal is observed from either analyte. By the time the pH has become basic, the polymer signal is characteristic of a neat, or non-matrix-assisted, desorption.
As shown with the above series of spectra, the sample and solution conditions must be optimized for the best performance of the matrix. We are continuing research on the effects of metal cations and other species on the performance of the matrix materials used in MALDI.
Further details about the effects of matrix pH and cationization on the MALDI analysis of polymers can be found in the publication:
![[*]](images/sphere02.gif) D. Dogruel, R.W. Nelson,
P.Williams, Rapid. Commun. Mass Spectrom. 1996, 10, 801-804
D. Dogruel, R.W. Nelson,
P.Williams, Rapid. Commun. Mass Spectrom. 1996, 10, 801-804
![[ <- ]](images/docsleft.gif) Back to Lab Page
Back to Lab Page  pwilliams@asu.edu Peter Williams
pwilliams@asu.edu Peter Williams