|
Problem statement
|
Watch Video
|
|
Force pushing atoms together
From lecture notes
|
 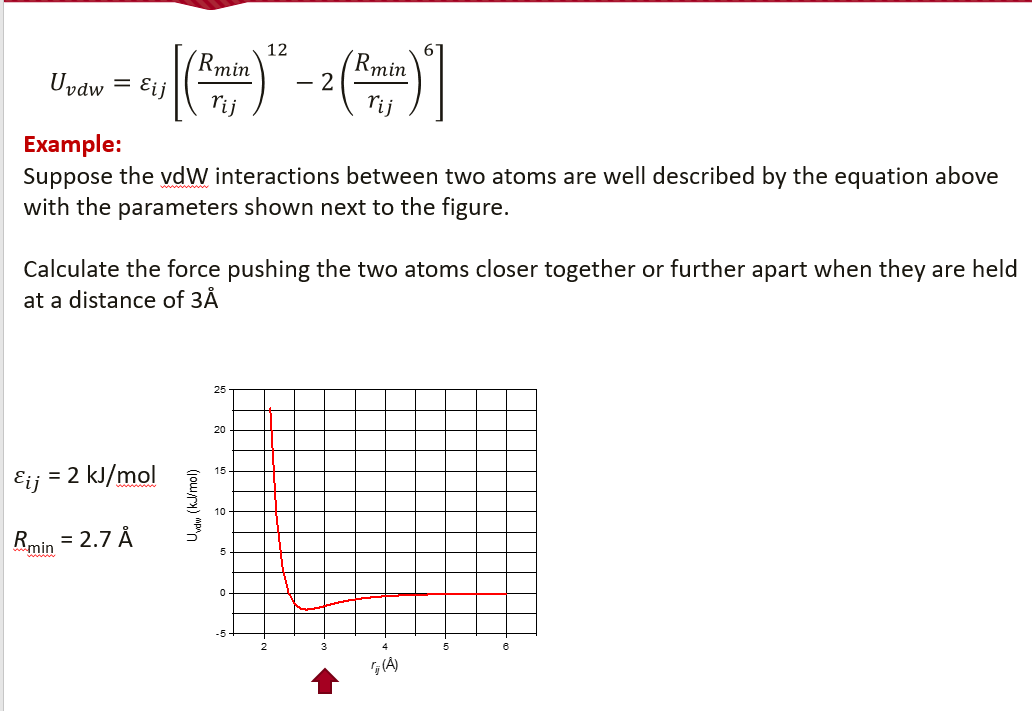
|

|
1
|
Work by a variable force
From lecture notes (Set 2, slide 32)
|
|

|
2
|
PV work
Use w=–Pex ΔV to calculate work
|
 
|

|
2
|
PV work Example 2
Reversible heating of a gas at constant pressure.
|
 
|

|
2
|
PV work
Follow up from lecture notes. Discusses how to avoid rounding errors.
|
|

|
2
|
q, w, ΔU and ΔH of reversible heating of an ideal gas at constant pressure
Tinoco's textbook. Chapter 2, problem 7.
|
 
|

|
2
|
ΔU and ΔH of a reaction from tabulated ΔH values of formation
Uses Cp data to calculate ΔU and ΔH at another temperature.
|
 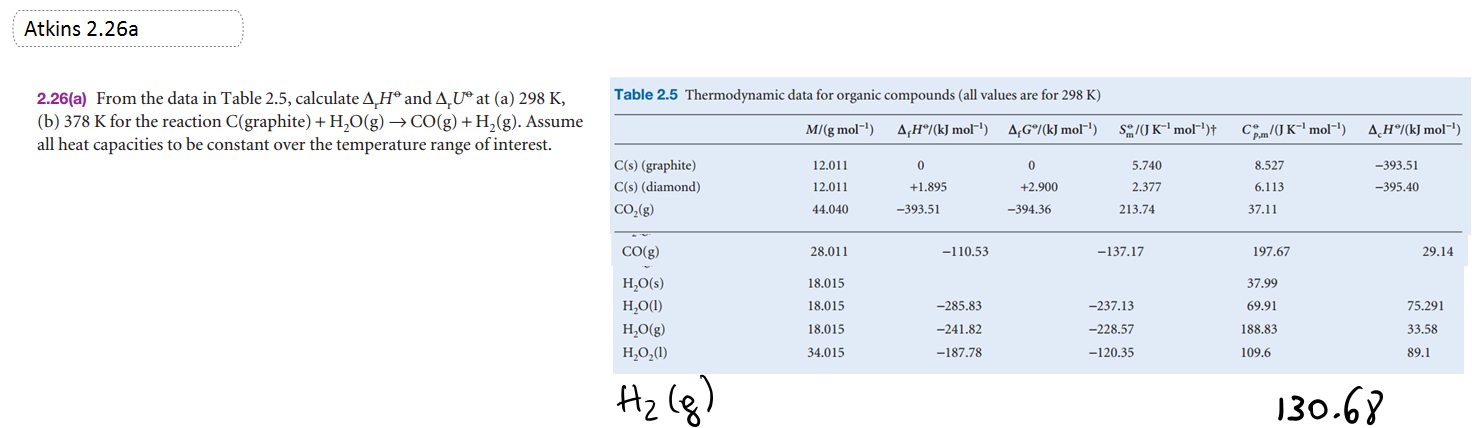
|

|
3
|
ΔS and ΔH of a reaction from tabulated data
Uses Cp data to calculate ΔS and ΔH at another temperature.2020 STUDENTS: DISREGARD THE PART OF THE PROBLEM THAT ASKS YOU ABOUT CHANGE OF ENTROPY AT A TEMPERATURE DIFFERENT THAN 298K. The part of the problem that discusses ΔH is part of HW3. The part about entropy is part of HW4.
|
 
|

|
3 4
|
ΔS of mixing hot and cold water.
|
 
|

|
4
|
ΔG of ATP hydrolysis.
|
|

|
5
|
Calculation of ΔG°' from ΔG°.
|
|

|
5
|
Protein-Ligand binding equilibrium
|
|

|
6
|
pH of ammonium chloride (2020 students: Remember that you can solve systems of equations in your computer. This recording was prepared before we went online)
|
 
|

|
7
|
pH of sodium acetate (2020 students: Remember that you can solve systems of equations in your computer. This recording was prepared before we went online)
|
 
|

|
7
|
pH of ammonium chloride (2020 students: Remember that you can solve systems of equations in your computer. This recording was prepared before we went online)
|
 
|

|
7
|
First order kinetics
|
|

|
10
|
Activation Energy
|
|

|
10
|