Adverse Outcome Pathways (AOPs) for Toxicology and Risk Assessment
As chemical toxicity testing moves toward the use of in vitro and shorter-term tests to evaluate chemicals more quickly and replace/reduce the number of animals used therein, mathematical/computational models will be needed to extrapolate results of those tests and predict effects in whole organisms. An adverse outcome pathway (AOP) spans multiple scales of biological organization from a molecular initiating event (e.g., enzyme inhibition or ligand-receptor binding) to effects on cells, organisms, or populations. An AOP describes qualitatively key events that are measureable and necessary for the adverse outcome to occur along with experimental evidence to support the key event and relationships between key events. A quantitative AOP (qAOP) is a mathematical model or set of models that predict the adverse outcome based due to perturbations in the AOP.
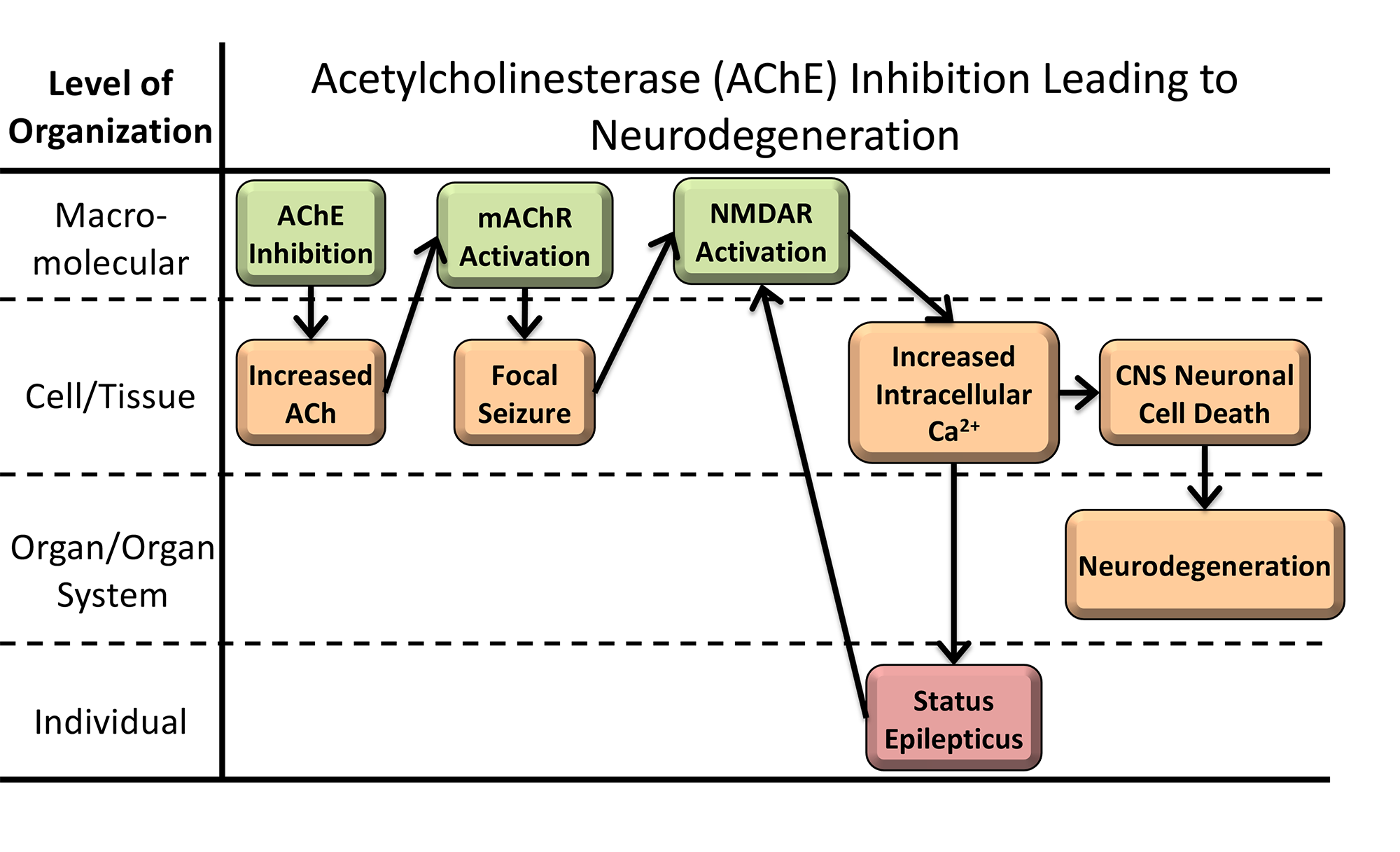
Acetylcholine regulates a wide variety of processes in our bodies such as muscle activation. An enzyme called acetylcholinesterase (AChE) eliminates acetylcholine, but some chemicals (e.g., pesticides and nerve agents) inhibit AChE. Too much acetylcholine causes a variety of adverse effects such as seizures, and paralysis. Our goal is to understand the range of sub-lethal adverse outcomes that arise due to AChE inhibition by developing a quantitative AOP (qAOP). The qAOP can be used to extrapolate changes measured in acetylcholinesterase activity and predict effects upon key events in the AOP. For a full description of the AOP visit the AOP Wiki. This research is supported by a cooperative agreement with the U.S. Army Corps of Engineers.
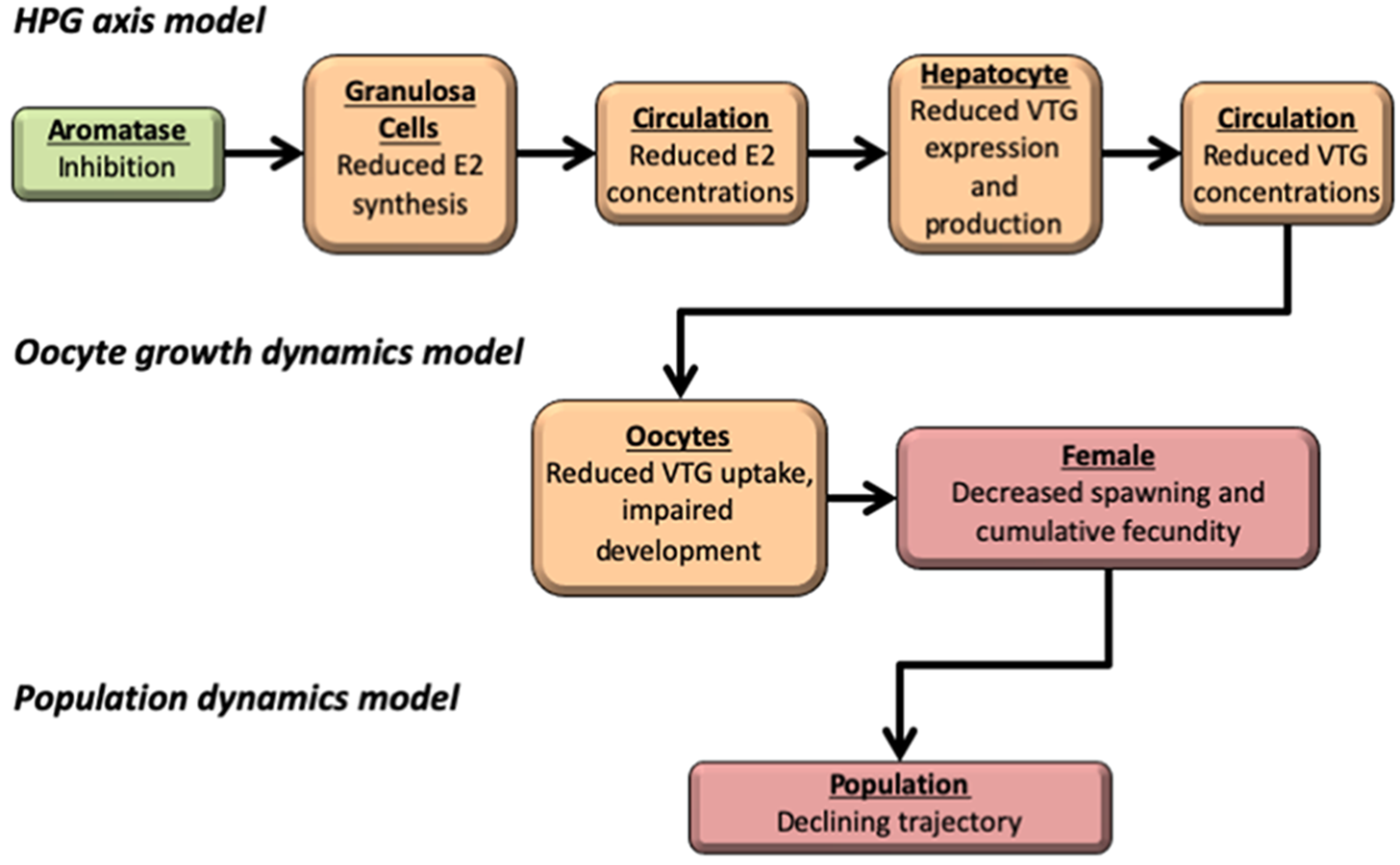
Aromatase is an enzyme that is needed to transform testosterone (an androgen) to estradiol (an estrogen). Some chemicals inhibit aromatase, and reduce the production of natural estrogens in many species. This qAOP represents how aromatase inhibitors affect fish reproduction and populations that are concerns for ecotoxicology and risk assessment. This qAOP comprises three independent mathematical models that are linked through their input and output values. Our goal is to develop predictive, multiscale models that represent the uncertainty and biological variability observed in fecundity data. This research was supported in part by a cooperative agreement with the U.S. Environmental Protection Agency.
NIMBioS workgroup, "Modeling Molecules to Organisms." Living organisms require energy to maintain biological process and grow or reproduce. However, biologically based mathematical models rarely account for energy requirements (e.g., food) in addition to chemical effects on outcomes such as reproduction. Our workgroup integrated Dynamic Energy Budget theory with quantitative AOPs for chemical effects upon a variety of endpoints such as growth and reproduction in fish. A case study focused on estrogen effects upon reproduction in salmon and demonstrated a method to integrate these two modeling approaches. To our knowledge, this was the first attempt to bring these two modeling fields together. This research was supported in part by the National Institute for Mathematical and Biological Synthesis (NIMBioS).